Contact : +91-93104 85979 / +91-95601 02589 / +91-98100 68241
Contact : +91-93104 85979 / +91-95601 02589 / +91-98100 68241
Life Sciences Sector Skill
Development Council Program
CPD Accredited
Post Graduate Diploma
Fulltime - 3 Months Live Online eLearning
Batch Commencement - Monday, 20th July 2026
Evening Sessions
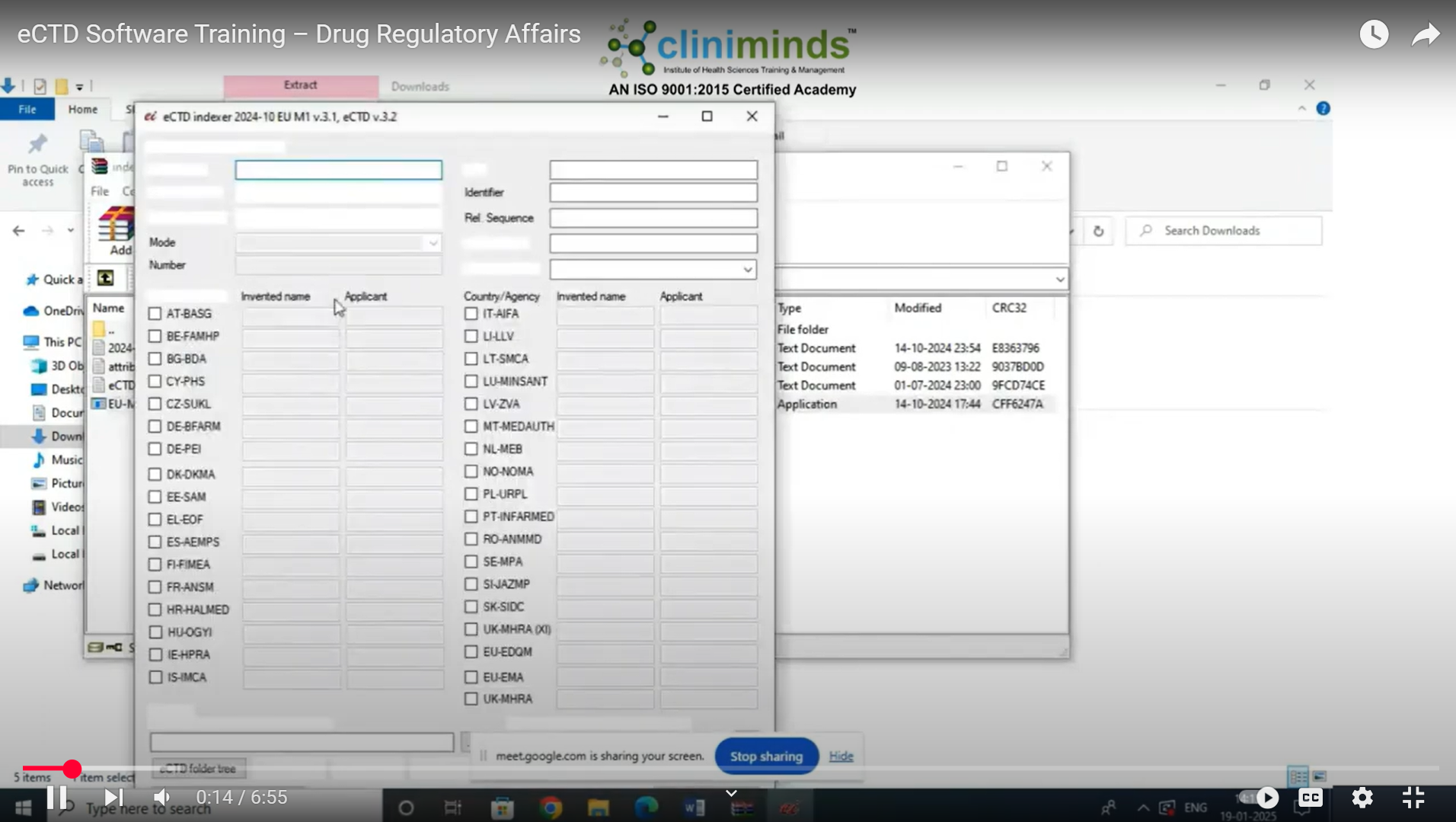
Specialized Training on eCTD Software
Building the Future of a Skilled
Life Sciences Workforce Since 2004
Cliniminds is now an Institutional Member
of the Indian Pharmaceutical Association (IPA) !
12,000+ Cliniminds Alumni
Eligibility - B.Pharmacy, M.Pharmacy, Doctor of Pharmacy, BSC, MSC, BDS, BHMS, BAMS, B.Tech / M.Tech Biotech, Biomedical, Life Sciences.
Life Sciences Sector Skill Development Council Program
CPD Accredited Post Graduate Diploma
Fulltime - 3 Months Live Online eLearning
Batch Commencement - Monday, 20th July 2026
Evening Sessions
Specialized Training on eCTD Software
Building the Future of a Skilled Life Sciences Workforce Since 2004
Cliniminds is now an Institutional Member of the
Indian Pharmaceutical Association (IPA) !
12,000+ Cliniminds Alumni
Eligibility - B.Pharmacy, M.Pharmacy, Doctor of Pharmacy, BSC, MSC, BDS, BHMS, BAMS, B.Tech / M.Tech Biotech, Biomedical, Life Sciences.
Program Inclusions
- Live Interactive eLearning Sessions
- Hands-on Software Training – eCTD
- Assured Job Interviews
- Dedicated Placement Cell - 360° Placement Support
- Job Role Specific Training
- Pre-Placement Training - CV Writing, Mock Interviews, English Communication, and other job-seeking skills
- Access to Learning Management System (LMS) – 24x7 - Recordings, PPTs, Notes, Modules
- Since 2004 – Over 12,000+ Placed Students
- Personalized Placement Support | Exclusive Job Opportunities
Certifications
-
1Life Sciences Sector Skill Development Council.
-
2Advanced Post Graduate Diploma – Cliniminds - CPD Continuing Professional Development. International Accreditation. Global Recognition
-
3Internship Certification – www.thinki.in
Certifications
-
1Life Sciences Sector Skill Development Council.
-
2Advanced Post Graduate Diploma – Cliniminds - CPD Continuing Professional Development. International Accreditation. Global Recognition
-
3Internship Certification – www.thinki.in
Module 1
- Drug Development Process & Introduction to Clinical Research
- Introduction to Regulatory Affairs, Terms & Terminologies & Abbreviations
- Chemistry, Manufacturing & Controls (CMC)
- Drug Master File
- Regulatory, Filing System for DMF in Different Countries
- ICH – Introduction, Objectives & Guidelines
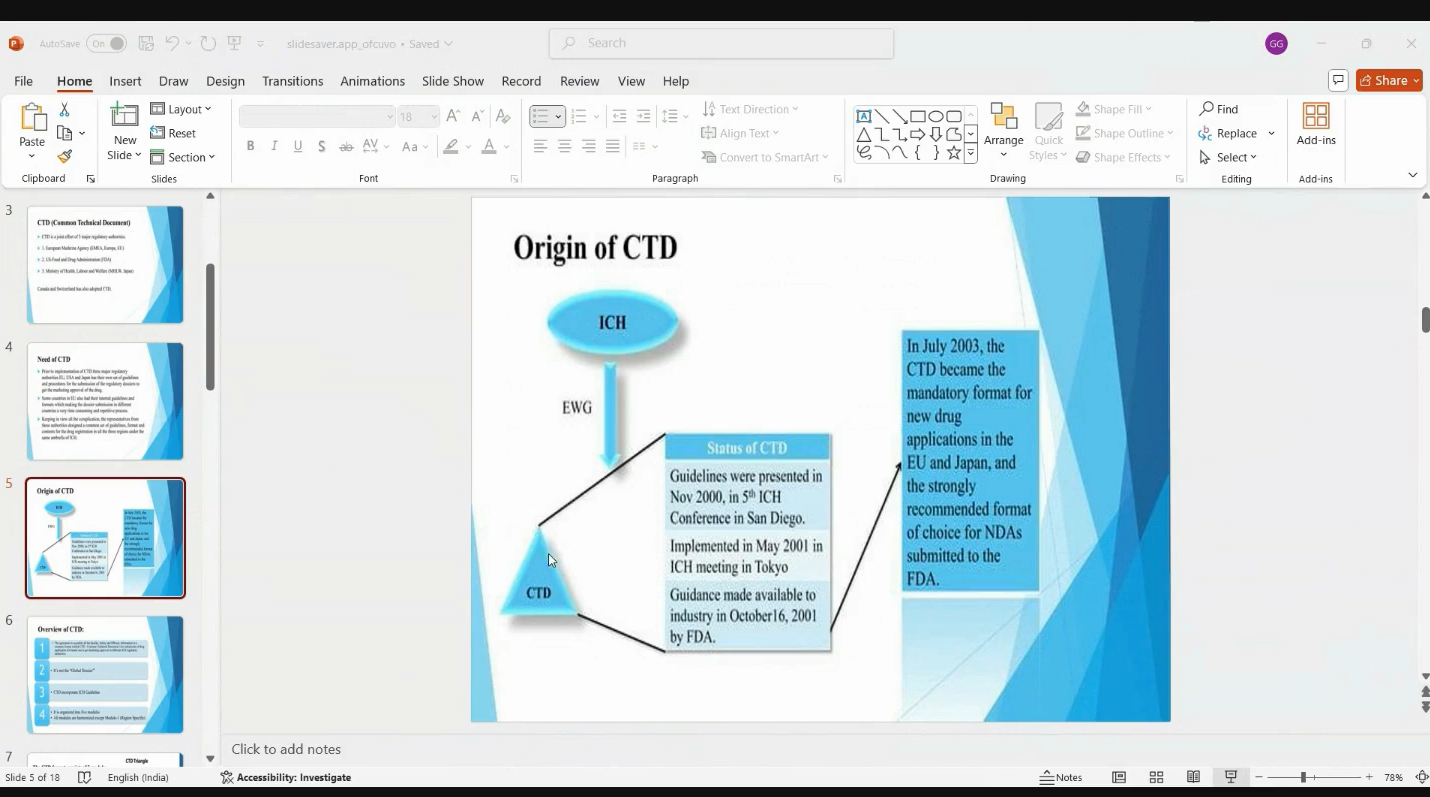
- CTD Introduction - Module 1 to 5
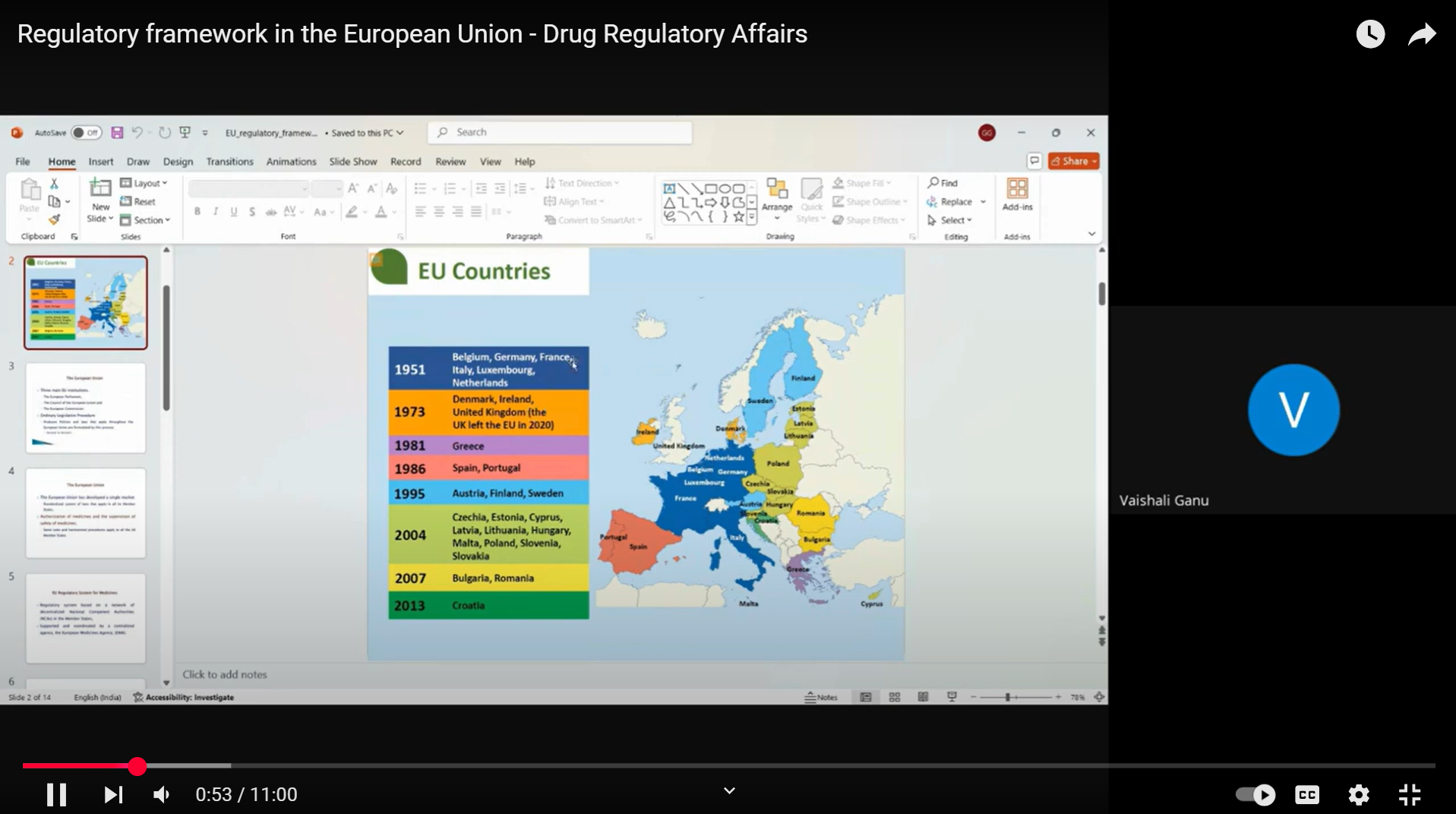
- Regulatory Filing Systems in Europe
- Regulatory Filing Systems in US
- Registration Procedures in Rest of the World
- Validations
- Audits & Compliances
- Regulatory Operations
- Pre Publishing
- Publishing
- Final Submission via Gateways
- Practical Approach towards Overall Dossier Preparation & Submission
- Pharmacovigilance Regulations
- Indian Pharmaceutical Regulations
Module 2
-
Essential Intellectual Property and Patent Management
-
Fundamentals of Intellectual Property (IP)
- Introduction to the IP Regime: Patents, Trademarks, Copyrights, Industrial Designs, Trade Secrets, Geographical Indications
-
Patent Essentials
- Understanding Patent Types and Patentability
- Anatomy of a Patent: Subject Matter, Inventorship, Term, and Expiry
-
Patent Application Process
- Disclosure Duties, Timelines, and Search Strategies
- Drafting Patents: Specification, Description, and Claims
-
Fundamentals of Intellectual Property (IP)

Introduction
This article has been written by the Regulatory Team at Cliniminds – www.cliniminds.com. The purpose of this article is to create awareness about the career opportunities in drug and medical device regulatory affairs.
For fresh graduates in pharmacy, life sciences, and biotech, the world of regulatory affairs (RA) offers a dynamic and rewarding career path. Regulatory affairs professionals play a crucial role in ensuring that pharmaceutical and medical device products are safe, effective, and compliant with all regulatory standards. This article aims to provide a comprehensive overview of careers in drug and medical device regulatory affairs, including the roles and responsibilities of regulatory professionals, the skills required, the types of submissions and processes, the importance of outsourcing RA services, various job roles, salary expectations, and key recruiters in the field.
What is Drug and Medical Device Regulatory Affairs?
Drug and medical device regulatory affairs involve managing the processes required to bring pharmaceutical and medical device products to market and ensuring they comply with all applicable regulations throughout their lifecycle. Regulatory professionals work to ensure that products meet all legal and scientific standards from initial development through to post-market surveillance.
Regulatory affairs encompass a wide range of activities including preparing and submitting regulatory documents, interfacing with regulatory agencies, ensuring compliance with regulatory requirements, and keeping up-to-date with changes in regulations. This field is vital for ensuring that safe and effective products are available to patients and consumers.
What Do Regulatory Professionals Do in Pharma and Medical Device Regulatory Affairs?
Regulatory professionals in the pharmaceutical and medical device industries have varied and essential roles, including:
- Regulatory Strategy Development: Developing regulatory strategies to guide product development and ensure compliance with all relevant regulations.
- Submission Preparation: Preparing and submitting regulatory documents to regulatory authorities such as the FDA, EMA, and others.
- Regulatory Compliance: Ensuring that all products and processes comply with current regulations and standards.
- Liaison with Regulatory Authorities: Communicating with regulatory agencies to discuss regulatory requirements, address queries, and provide additional information as needed.
- Post-Market Surveillance: Monitoring products after they have been launched to ensure ongoing compliance and addressing any regulatory issues that arise.
- Regulatory Intelligence: Keeping abreast of changes in regulations and guidelines that may impact product development and marketing.
Key Technical and Job Skills Required for Pharma and Medical Device Regulatory Professionals
To excel in regulatory affairs, professionals need a mix of technical and job-specific skills, including:
- Technical Knowledge: Understanding of the scientific and technical aspects of drug and medical device development.
- Regulatory Knowledge: Familiarity with regulatory guidelines, standards, and submission requirements.
- Attention to Detail: Precision in preparing and reviewing regulatory documents to ensure accuracy and compliance.
- Communication Skills: Strong written and verbal communication skills to interact effectively with regulatory authorities and cross-functional teams.
- Project Management: Ability to manage multiple projects simultaneously and meet deadlines.
- Problem-Solving Skills: Analytical skills to address regulatory issues and find compliant solutions.
- Adaptability: Ability to adapt to changing regulations and evolving industry standards.
Types of Pharma and Medical Device Regulatory Submissions and Processes
Understanding the various types of regulatory submissions and processes is crucial for students entering this field. Key types include:
- Investigational New Drug (IND) Application: Required for the initiation of clinical trials for new drugs.
- New Drug Application (NDA): Submitted for approval to market a new drug.
- Biologics License Application (BLA): Required for the approval of biological products.
- Abbreviated New Drug Application (ANDA): Submitted for the approval of generic drugs.
- Premarket Approval (PMA): Required for high-risk medical devices.
- 510(k) Submission: Required for demonstrating that a medical device is substantially equivalent to a legally marketed device.
- Clinical Trial Applications (CTA): Necessary for conducting clinical trials in various regions.
- Periodic Safety Update Reports (PSURs): Required for ongoing post-market surveillance and safety reporting.
Why Pharma and Medical Device Companies Outsource Regulatory Affairs Services
Pharmaceutical and medical device companies often outsource regulatory affairs services for several reasons:
- Expertise: Access to specialized knowledge and expertise in regulatory requirements and processes.
- Cost-Effectiveness: Reducing the costs associated with maintaining an in-house regulatory affairs team.
- Efficiency: Accelerating the regulatory submission and approval process by leveraging the experience of regulatory service providers.
- Flexibility: Ability to scale regulatory activities up or down based on project needs.
The market for outsourcing regulatory affairs services is growing, driven by the increasing complexity of regulations and the need for specialized expertise. According to industry reports, the global regulatory affairs outsourcing market is expected to reach USD 2.73 billion by 2025, growing at a CAGR of 11.9%.
Job Roles in Regulatory Affairs Department
Regulatory affairs departments in pharmaceutical and medical device companies offer various job roles, including:
Entry-Level Roles:
- Regulatory Affairs Associate
- Regulatory Affairs Specialist
Middle Management Roles:
- Regulatory Affairs Manager
- Senior Regulatory Affairs Specialist
Senior Management Roles:
- Director of Regulatory Affairs
- Vice President of Regulatory Affairs
Career growth in regulatory affairs can be significant, with opportunities to advance to senior leadership positions through gaining experience, expertise, and additional qualifications.
Salary Expectations in Regulatory Affairs
Salaries in regulatory affairs can vary based on experience, qualifications, and the specific role. Estimated annual salaries in India are:
- Entry-Level Roles: INR 4-8 lakhs
- Middle Management Roles: INR 10-18 lakhs
- Senior Management Roles: INR 18 – 30 lakhs or more
Salary growth in regulatory affairs is robust, with professionals typically seeing significant increases as they advance in their careers.
Key Global and Indian Recruiters for Regulatory Affairs Jobs
Several global and Indian pharmaceutical and medical device companies, as well as consulting firms, are key recruiters for regulatory affairs professionals:
Global Recruiters:
- Pfizer
- Roche
- Novartis
- GlaxoSmithKline
- Sanofi
- Johnson & Johnson
- Merck
- AstraZeneca
- Bayer
- AbbVie
- Amgen
- Boehringer Ingelheim
- Eli Lilly and Company
- Gilead Sciences
- Teva Pharmaceutical Industries
Indian Recruiters:
- Dr. Reddy's Laboratories
- Cipla
- Sun Pharmaceutical Industries
- Biocon
- Glenmark Pharmaceuticals
- Lupin
- Wockhardt
- Cadila Healthcare (Zydus Cadila)
- Torrent Pharmaceuticals
- Aurobindo Pharma
- Alkem Laboratories
- Jubilant Life Sciences
- Piramal Enterprises
- Natco Pharma
- Intas Pharmaceuticals
- Hetero Drugs
- Alembic Pharmaceuticals
- Strides Pharma Science
Consulting Firms:
- Freyr
- IQVIA
- PAREXEL
- Covance
- ICON plc
- Syneos Health
- Charles River Laboratories
- Medpace
- PRA Health Sciences
- Pharmalex
- Kinapse
Conclusion
A career in drug and medical device regulatory affairs offers a dynamic and rewarding path for pharmacy, life sciences, and biotech graduates. By understanding the roles and responsibilities of regulatory professionals, the skills required, and the various types of regulatory submissions and processes, students can better prepare for successful careers in this field. With opportunities for career growth, competitive salaries, and the increasing demand for regulatory expertise, regulatory affairs is an excellent choice for those interested in ensuring the safety and efficacy of pharmaceutical and medical device products.
References
- EMA Guidelines
- FDA Regulatory Submissions
- "Global Regulatory Affairs Outsourcing Market" Report
- Industry salary surveys and job portals
Cliniminds Awarded Institution with Excellent Training & Placements at Business World Top Education Brands Awards
Cliniminds has entered into the agreement with Life Sciences Sector Skill Development Council (LSSSDC) under the Ministry of Skill Development & Entrepreneurship.
Cliniminds – www.cliniminds.com – Established in 2004, offers life sciences sector job oriented, up skilling and professional training programs in the areas of clinical trials, pharmacovigilance, data management, business analytics, medical writing, regulatory affairs and other specialized areas. Cliniminds has an alumni of over 11,500+ professionals from India, US, Europe, Middle East, Africa and Latin America. Cliniminds programs are accredited and certified by Life Sciences Sector Skill Development Council, Ministry of Skill Development & Entrepreneurship and CPD Continuing Professional Development, International Accreditation.
Cliniminds Awarded Best Pharma Skilling Institute of India by PHD Chamber of Commerce and Industry
This program offers a Certificate Program in Drug Regulatory Affairs, IPR & Patent, in addition to the Cliniminds CPD Accredited Post Graduate Diploma provided by Cliniminds. The LSSSDC certification will enhances the value of your qualifications and also significantly boosts your prospects for successful job placements.
Cliniminds has seasoned senior industry experienced faculty, experienced placement team, robust IT platform, ERP/LMS, and business development teams. Cliniminds has strong international foot print has active students form 20+ countries, including developed markets like US and Europe. Cliniminds has recently singed the MoU with the leading Management & Science University, Malaysia to offer Cliniminds training program at their Malaysian campus. Cliniminds provides corporate training solutions to the leading corporates like Oracle, Birla Soft, Ultragenic, Bio Pharma, USA and many more.