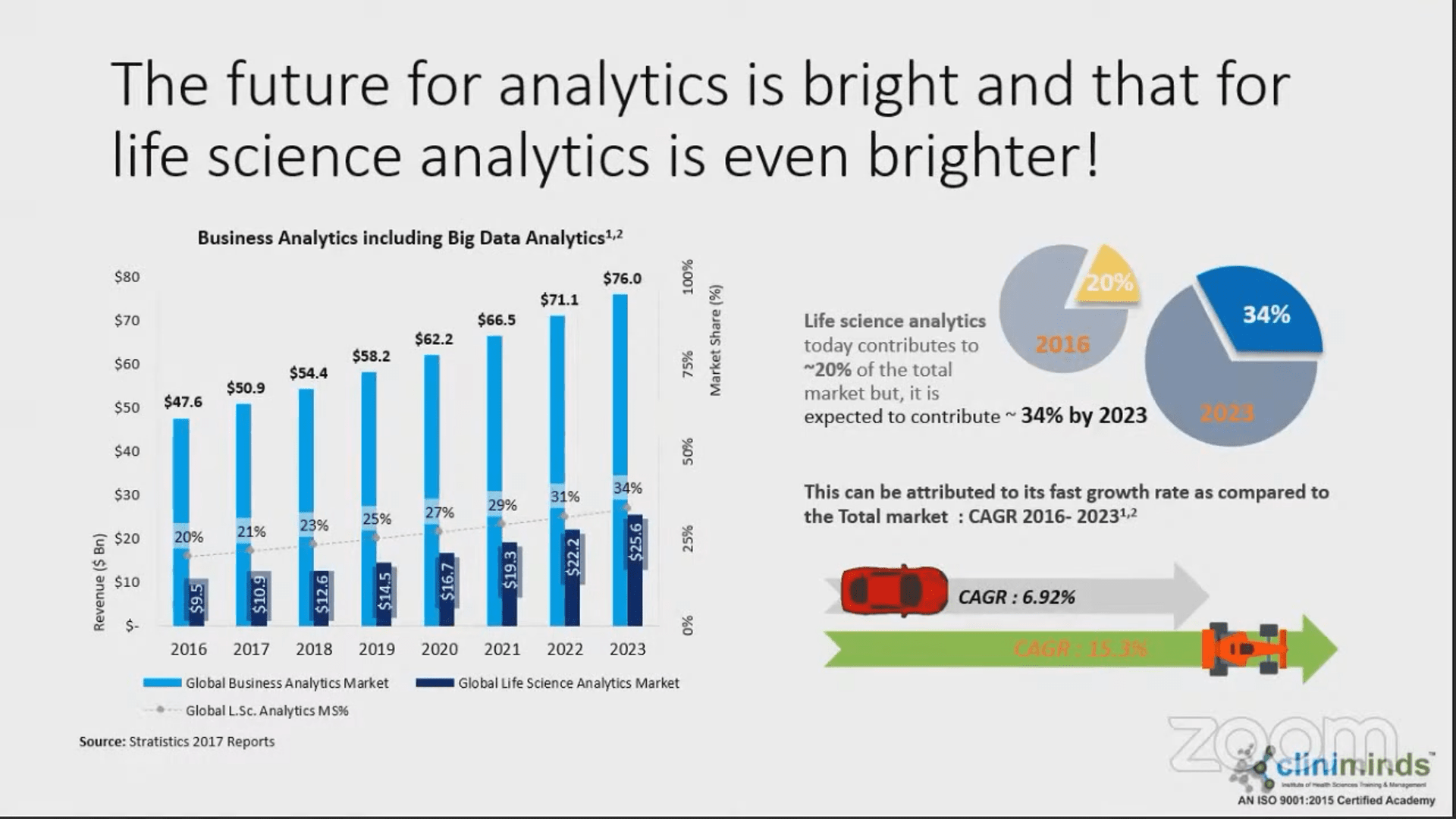
Previous Webinars
Youtube
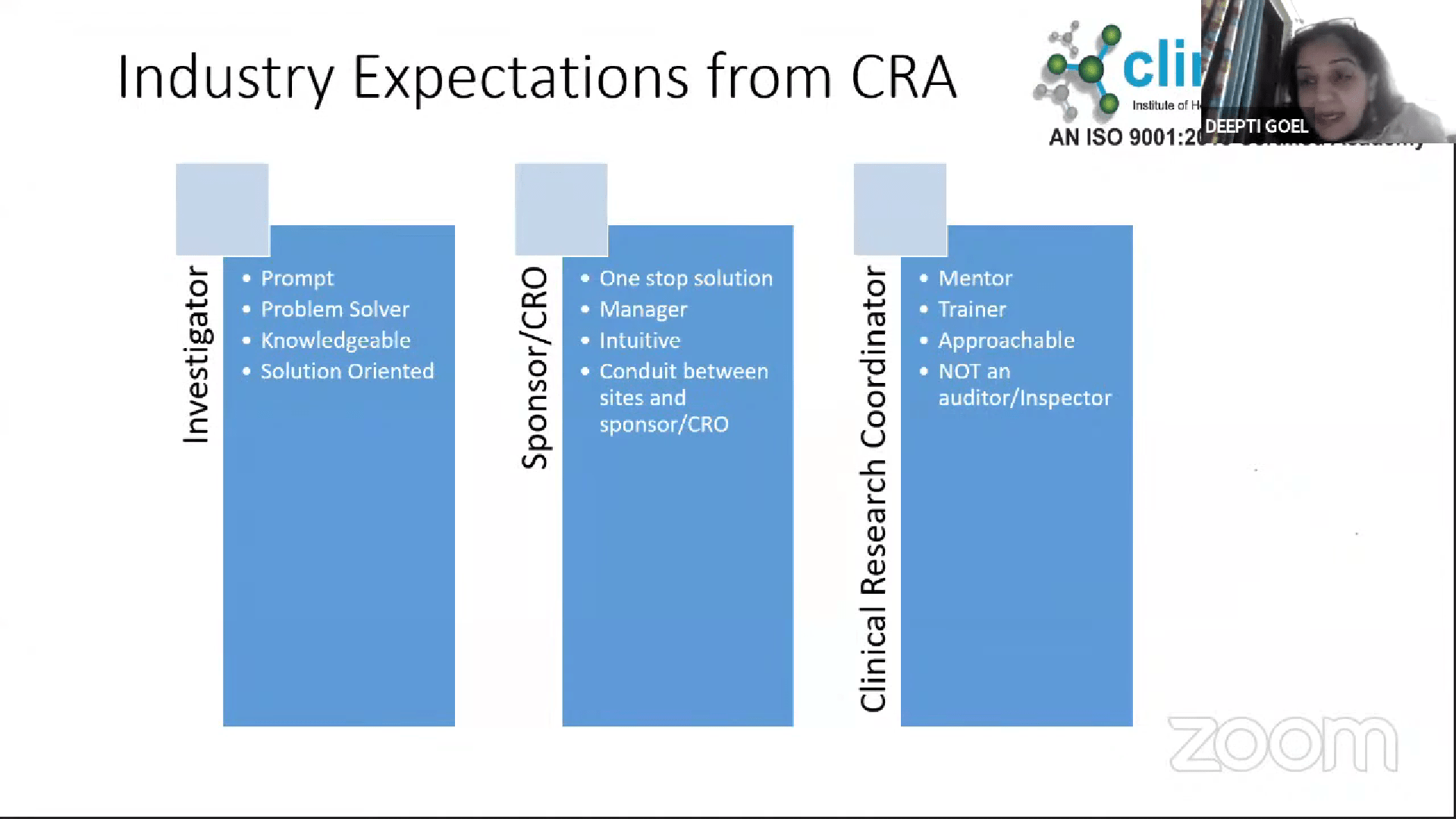
Webinar Recent Technological Advances in Reproductive Medicine & Ultrasound
7th February 2021
Youtube
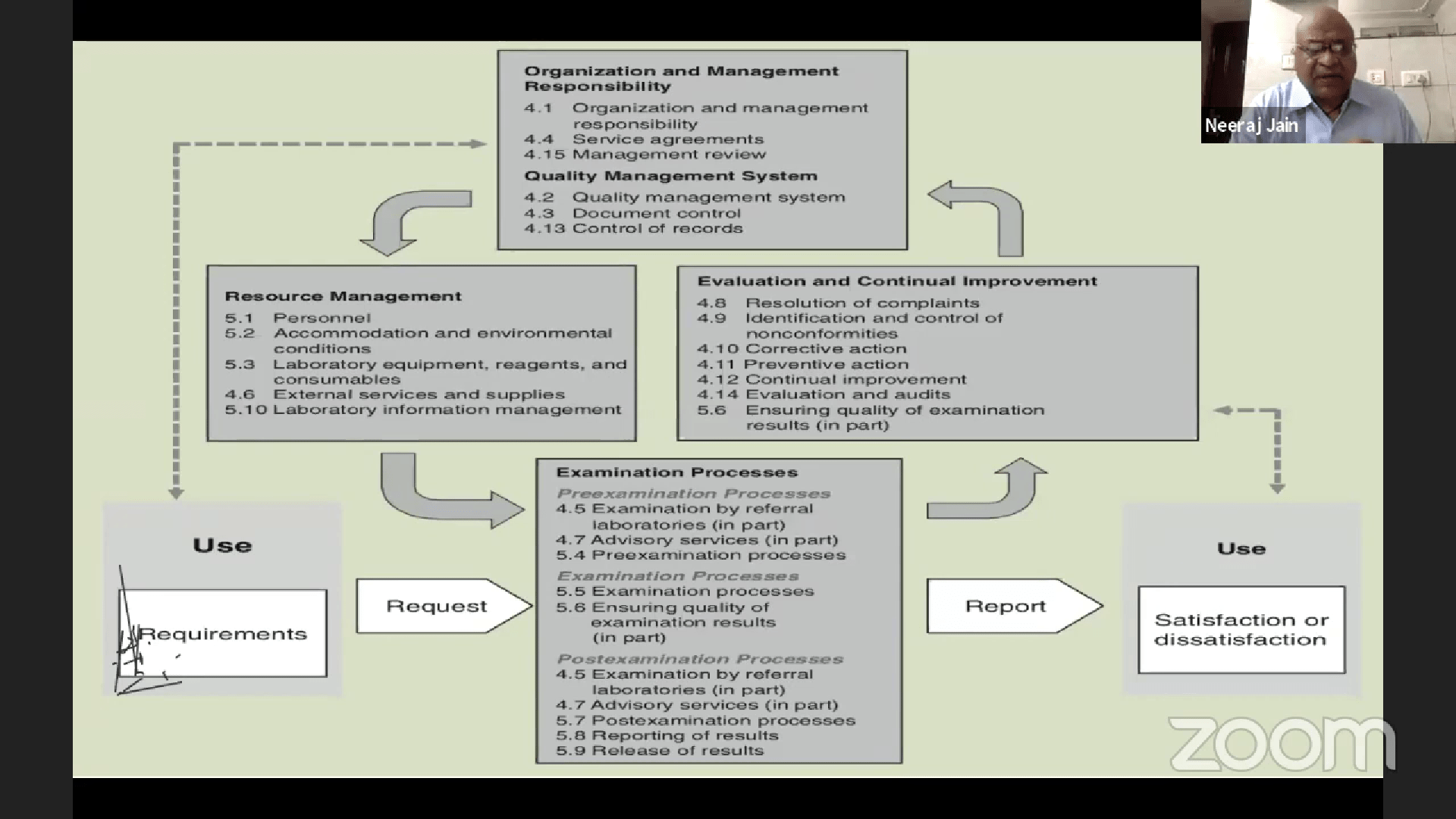
Impact of COVID19 on Clinical Laboratories and the role of Lab Accreditation
02nd August 2020